While it is well established that signal intensity and duration can lead to drastically different outcomes during development, its impact on tumorigenesis remains poorly characterized. In this study supervised by Damien Grégoire and Ula Hibner, our team studied such quantitative heterogeneity of signal intensity in the context of MAPK pathway activation in hepatocellular carcinoma (HCC), the most frequent primary liver cancer. We hypothesized that variations in the signaling intensities of the MAPK Erk, which is activated in 30-50% of HCC, might trigger selective pressures that generate functional heterogeneity in otherwise genetically identical tumor cells.
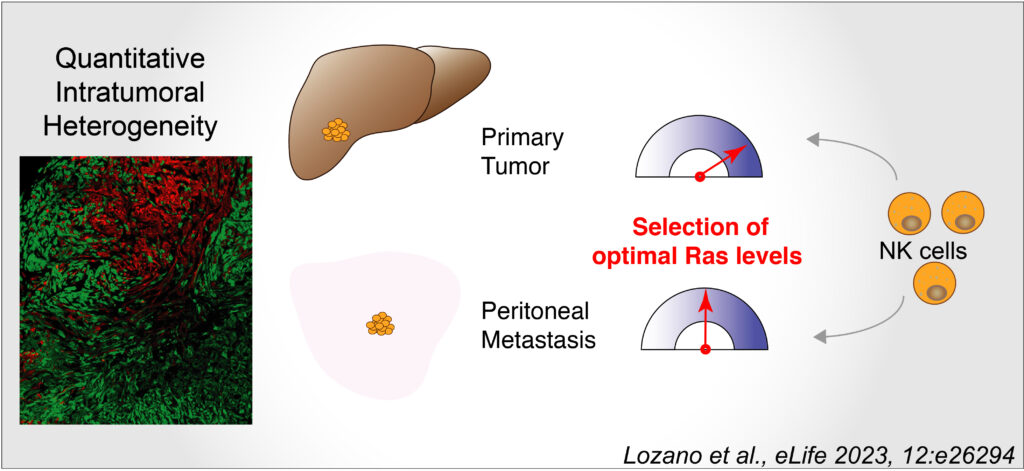
In order to study population dynamics and intercellular interactions during tumor growth and dissemination, we developed a murine model of HCC that combines intrahepatic xenografts and lineage tracing. To model the activation of MAPK we used retroviral transduction of a constitutively active form of the Ras oncogene – HRasG12V. The use of a bi-cistronic vector expressing fluorescent reporter proteins allowed the determination of oncogene dosage, which was proportional to fluorescence intensity in individual cells. Populations of cells expressing defined range of the oncogene were purified by cell sorting, characterized ex vivo and injected as orthotopic allografts.
Our experimental model gave rise to both primary tumors and extrahepatic growth. We showed that strong selective pressures operate in both tumoral locations, giving rise to tumors expressing an optimal, narrow ranges of the oncogene dosage, reflective of MAPK Erk signaling activity. Strikingly, the signal intensities compatible with tumorigenesis at the primary and metastatic sites were significantly different. Indeed, significantly higher Ras expression was observed in primary as compared to metastatic tumours, despite the evolutionary trade-off of increased apoptotic death in the liver that correlated with high Ras dosage. Functionally, we identified several components of the immune stroma that participate in the definition of cellular fitness in these two host microenvironments, among which NK cells appear to play a key role in defining the selective milieu controlling tumor growth dynamics.
Overall, our work describes a quantitative aspect of tumour heterogeneity and highlights a potential vulnerability of a subtype of hepatocellular carcinoma as a function of MAPK Erk signaling intensity.
https://elifesciences.org/articles/76294
Lozano A., Souche FR., Chavey C., Dardalhon V., Ramirez C., Vegna S., Desandré G., Riviere A., Zine El Aabidine A., Fort P., Akkari L., Hibner U. *, Grégoire D.* (2023) Ras/MAPK signalling intensity defines subclonal fitness in a mouse model of hepatocellular carcinoma. eLife, 2023 12:e76294. https://doi.org/10.7554/eLife.76294